Jumo Health and Areti Health Announce Strategic Partnership to Advance Readiness-Based Clinical Trial Execution
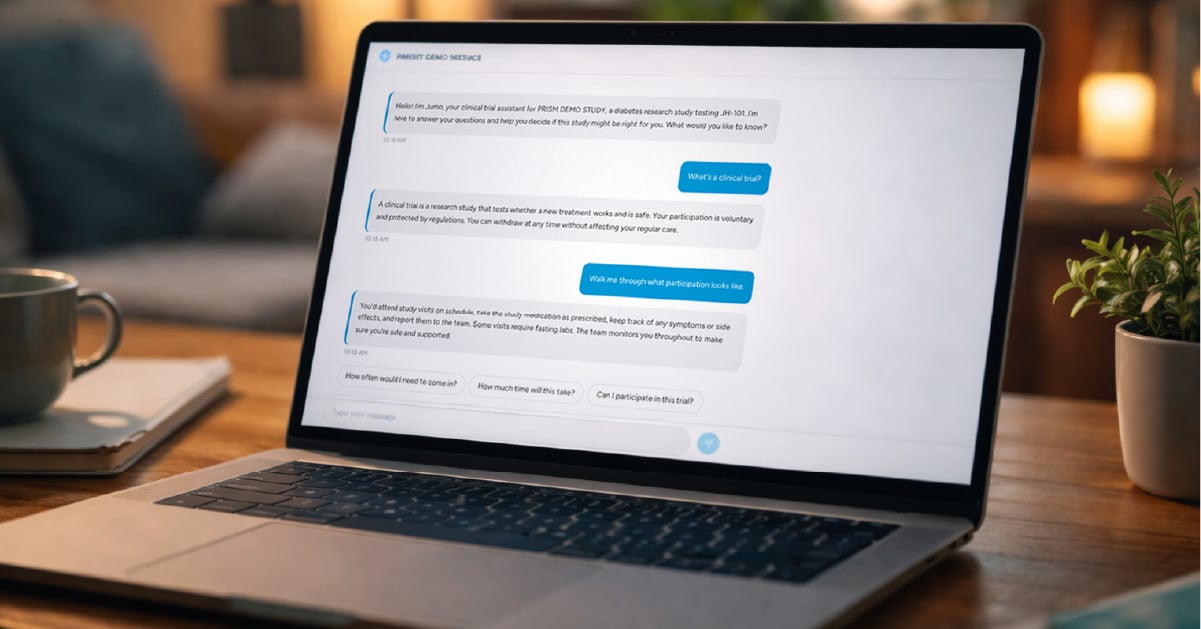
Partnership strengthens patent pending PRISM™, Jumo Health's readiness-based recruitment, enrollment, and retention system, with advanced AI delivery...